Honsin Apparel Sdn. Bhd., a wholly-owned subsidiary of Prolexus Berhad (Prolexus), is registered with the U.S. Food and Drug Administration (FDA) for face mask for general public / healthcare personnel per Immediately in Effect Guidance (IIE Guidance). The registration is expected to expand ProXmask™ export market horizon to the US market to curb Covid-19 pandemic.
Prolexus, which is already an established international sportswear manufacturer, has aggressively responded to the unprecedented pandemic crisis through the development of ProXmask™ as one of its core products. Prolexus aspires to deliver high quality reusable functional fabric mask to ensure effective protection for everyone against the virus.
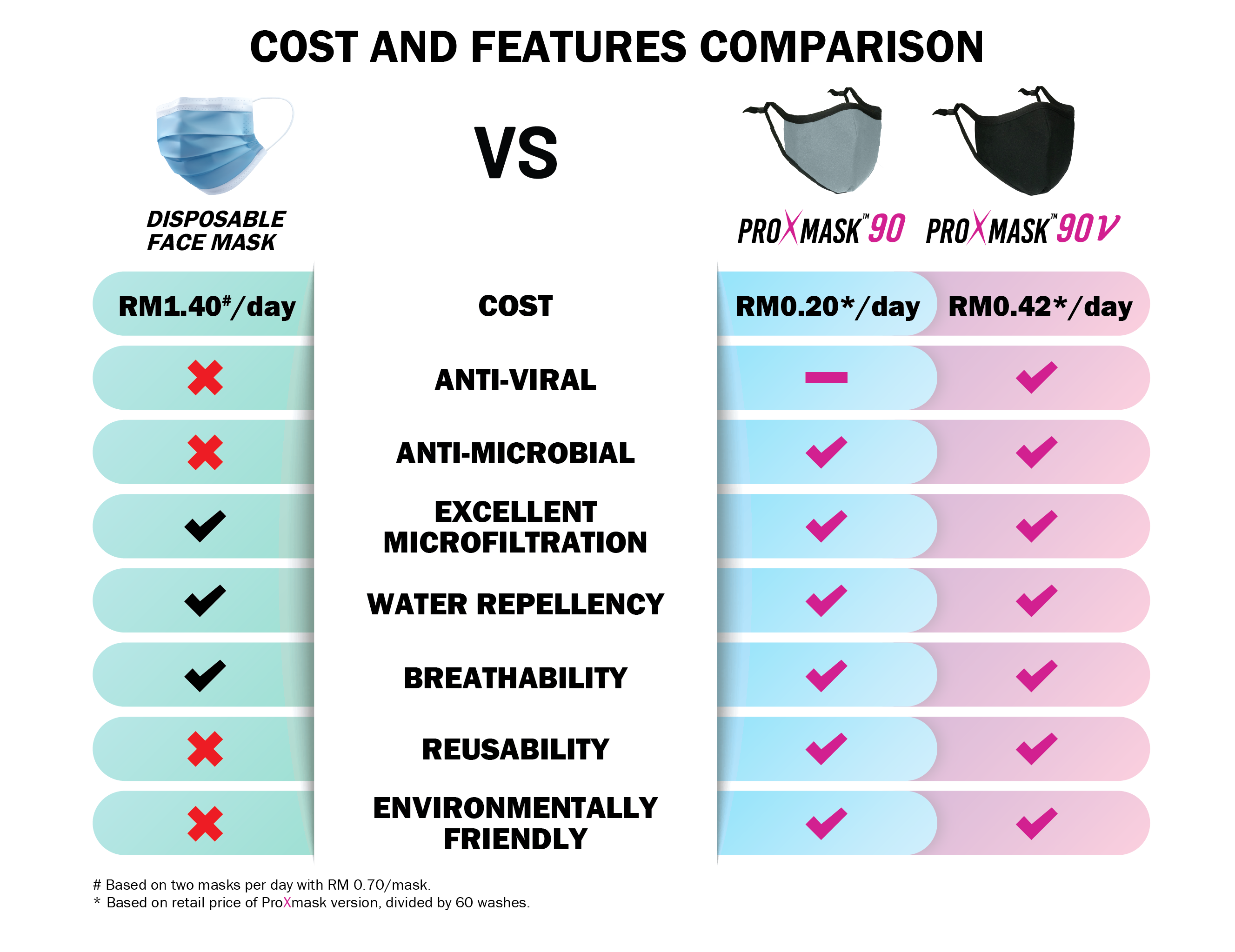
Prolexus revealed its cutting edge ProX™ anti-virus technology which inactivates bacteria and viruses, including >99% SARS-CoV-2, the virus responsible for Covid-19. The technology, accompanied with its water repellency and microfiltration properties, enables ProXmask™ to be both protective and preventive, benefiting both self and others in close proximity. The absence of self-disinfection property in conventional disposal masks however, increases the risk of disease spread upon improper handling and disposal.
Moreover, the reusability of ProXmask™ makes it an eco-friendly and cost-effective choice. It stays effective up to 60 wash cycles, making it equivalent to at least 120 disposal masks, assuming a 4 to 6 hours of usage lifespan (as doctors' advise) per disposal mask. Its protective features also exceed the ones in disposal masks, making it a value for money product.
ProXmask™ protective features have passed the test conducted by various international accredited laboratories. Backed by its advanced ProX™ anti-virus technology, Prolexus is set to explore innovative market segments such as personal protective apparels, a new emerging sportswear category. Prolexus marketing is actively working towards serving prospective clients with interest in protective and functional activewears.
Any interested parties, please call 1700-81-8398, email [email protected] or visit www.prolexus.com.my/proxmask